|
Cite this article as: He J, Zhao F, Li H, Zhou K, Zhu B. Pancreatic paraganglioma: A case report of CT manifestations and literature review. Quant Imaging Med Surg 2011;1:41-43. DOI: 10.3978/j.issn.2223-4292.2011.08.02
Case Report
Pancreatic paraganglioma: a case report of CT manifestations and literature review
Jian He1, Feng Zhao2, Hui Li1, Kefeng Zhou1, Bin Zhu1*
1Department of Radiology, Nanjing Drum Tower Hospital, the Affiliated Hospital of Nanjing University Medical School, Nanjing, China; 2Department of Imaging and Interventional Radiology, the Chinese University of Hong Kong, Shatin, N.T., Hong Kong, China
Corresponding to: Bin Zhu, MD. Department of Radiology, Nanjing Drum Tower Hospital, the Affiliated Hospital of Nanjing University Medical School, 210008, No.321 Zhongshan Road, Nanjing, Jiangsu Province, China. Telephone number: 86-25-83105166; Fax number: 86-25-83105166. E-mail: gobincdr@gmail.com.
|
|
Abstract
Paragangliomas are rare tumors arising from paraganglia (or chromaffin bodies) within the ganglia of the sympathetic trunk and the ganglia of the celiac, renal, suprarenal, aortic and hypogastric plexuses. These tumors can be widely distributed from skull base to the bottom of pelvis. Paraganglioma originating from pancreas is extremely rare. Here we report a 40-year-old female patient with paraganglioma located in the pancreatic head as a solid, well-demarcated mass with substantial enhancement on contrast-enhanced CT examination. Relevant literatures are also reviewed.
Key words
paraganglioma; pancreas; computer tomography
Quant Imaging Med Surg 2011;1:41-43. DOI: 10.3978/j.issn.2223-4292.2011.08.02
|
|
Introduction
A paraganglioma is a rare neoplasm originating from paraganglia (or chromaffin bodies) within the ganglia of the sympathetic trunk and the ganglia of the celiac, renal, suprarenal, aortic and hypogastric plexuses. Although it is rare, it can be found in abdomen, pelvis, and the head and neck region. In abdominal region, most paraganglias are located at adrenal glands or along aorta (the latter is known as Zuckerkandl body). To our knowledge, only 12 cases of paraganglioma arising from pancreas have been reported in English literatures, most of which are in pathological literatures.
|
|
Case report
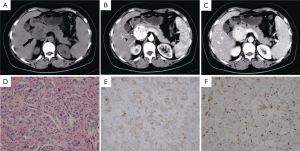
A pancreatic mass was revealed in a 40-year-old woman by abdominal ultrasonography at a routine health check. She did not complain any relevant symptoms. Her family history, past history, physical examination, and laboratory test were unremarkable, except slightly elevated serum CA15-3 (30.04 U/ml; normal, <25 U/ml). Abdominal CT showed a well-demarcated solid mass measured 4.5 cm×4.2 cm in the uncinate process of the pancreas ( Fig 1A). Contrast-enhanced CT showed that the tumor demonstrated dramatical enhancement at pancreatic and portal vein phases ( Fig 1B, C) . Several low-density foci were revealed also at plain and enhanced scans. Mild dilation of pancreatic duct was noted at the body and tail of pancreas. No biliary dilation or liver lesions were detected. The resected mass was grey-yellow in color, with multifocal hemorrhagic areas and cystic degeneration. Microscopically, the tumor was composed of nests of cells arranged in a classic Zellballen pattern separated by a delicate vascular network ( Fig 1D). The chief cells showed positive staining to neuron-specific enolase, chromogranin A, synaptophysin, and cytokeratin ( Fig 1E), but showed negative response to insulin, somatostatin, and glucagon. Less than 2% of the tumor cells were positive to Ki67, indicating a low proliferation ratio. The chief cells were surrounded by S-100 protein-positive sustentacular cells ( Fig 1F). Regional lymph nodes and the resected margins were free of tumor cells. A diagnosis of paraganglioma of the pancreas was established. |
|
Discussion
Paragangliomas originate from the neural crest cells that are widely located in adrenal medulla, carotid and aortic body, jugular foramen, middle ear, aortopulmonary region, the organ of Zuckerkandl, and other sites of the body. The adrenal gland is the most common primary site of paragangliamas, followed by priaorta region and the head and neck. Paragangliomas were also found in many other organs. Only 12 cases of pancreatic paragangliomas have been reported in English literatures,( 1- 10) and only two of them were focused on radiological mani-festations. In other words, the imaging features of pancreatic paragangliomas have not been well described. In our current case, the pancreatic paraganglioma appeared as a solid mass with necrotic foci or cystic changes on CT scans. Dramatical enhancement was found on contrast-enhanced CT. Although it shares similar imaging characteristics with other neuroendocrine neoplasms of pancreas, its CT findings are different from pancreatic carcinoma, which often appears as a poor enhanced mass with dilations of bile ducts and pancreatic duct as well as retroperitoneal involvement. Although the pancreatic paraganglioma is located at the head of pancreas, the biliary ducts usually show no dilation and patients show no jaundice, which are helpful in differentiating pancreatic paraganglioma from pancreatic carcinoma. Paragangliomas at various sites share relatively identical imaging features, including clear margin, hypervascularity, and cystic areas within the tumor; the latter demonstrates cystic degeneration or hemorrhagic necrosis. In our current case, these imaging characteristics were also found. Although most paragangliomas are solitary, 8 among 13 cases of pancreatic paragangliomas were cystic ( 1- 10). Cystic pancreatic paragangliomas ranged from 4.0 cm to 13.0 cm in diameter, with a median of 8.2 cm. Solid pancreatic paragangliomas ranged from 1.5 cm to 7.0 cm in diameter, with a median of 3.4 cm. Differential diagnosis between pancreatic cystic paraganglioma and pseudocyst can be difficult. According to literatures, four patients were misdiagnosed preoperatively and drainage was carried out in two of them ( 2, 3). It is also difficult to differentiate between solid paraganglioma and other neuroendocrine tumors of the pancreas, especially those nonfunctional pancreatic paragangliomas. Kim SY et al argued that early contrast filling of the prominent draining veins of solid paraganglioma and the portal vein might provide clues for differential diagnosis ( 4). However, prominent draining veins of solid paraganglioma were not observed in our patient. Actually, we observed plenty of slightly thicken peritumoral arteries and early contrast filling of the portal vein, which is believed to be a common phenomenon of hypervascular tumors of the pancreas. Occasionally, extra-adrenal paraganglioma arising from the retroperitoneum and abutting the pancreas may mimick a pancreatic neoplasm ( 11). In conclusion, we present a rare paraganglioma arising in pancreas, whose CT findings were featured by a well-demarcated solid mass with necrotic foci or cystic changes and by the dramatic enhancement after the intravenous administration of contrast media. These findings are similar with those of paragangliomas in other sites and other neuroendocrine neoplasm of pancreas, which may be helpful to urge the radiologists to consider the possibility of paraganglioma during differential diagnosis.
|
|
References
- Malthouse SR, Robinson L, Rankin SC. Ultrasonic and computed tomographic appearances of paraganglioma simulating pancreatic mass. Clin Radiol 1992;45:271-2.[LinkOut]
- Bartley O, Ekdahl PH, Hultén L. Paraganglioma simulating pancreatic cyst. Acta Chir Scand 1966;132:289-97.[LinkOut]
- Zamir O, Amir G, Lernau O, et al. Nonfunctional paraganglioma of the pancreas. Am J Gastroenterol 1984;79:761-3.[LinkOut]
- Kim SY, Byun JH, Choi G, et al. A case of primary paraganglioma that arose in the pancreas: the Color Doppler ultrasonography and dynamic CT features. Korean J Radiol 2008;9 Suppl:S18-21.[LinkOut]
- Fujino Y, Nagata Y, Ogino K, et al. Nonfunctional paraganglioma of the pancreas: report of a case. Surg Today 1998;28:209-12.[LinkOut]
- Goodof II, Lischer CE, Louis S. Tumor of the carotid body and the pancreas. Arch Pathol 1943;35:906-11.
- Cope C, Greenberg SH, Vidal JJ, et al. Nonfunctioning nonchromaffin paraganglioma of the pancreas. Arch Surg 1974;109:440-2.[LinkOut]
- Parithivel VS, Niazi M, Malhotra AK, et al. Paraganglioma of the pancreas: literature review and case report. Dig Dis Sci 2000;45:438-41.[LinkOut]
- Ohkawara T, Naruse H, Takeda H, et al. Primary paraganglioma of the head of pancreas: contribution of combinatorial image analyses to the diagnosis of disease. Intern Med 2005;44:1195-6.[LinkOut]
- Tsukada A, Ishizaki Y, Nobukawa B, et al. Paraganglioma of the pancreas: a case report and review of the literature. Pancreas 2008;36:214-6.[LinkOut]
- Lightfoot N, Santos P, Nikfarjam M. Paraganglioma mimicking a pancreatic neoplasm. JOP 2011;12:259-61.[LinkOut]
|