Comparison of dual energy subtraction chest radiography and traditional chest X-rays in the detection of pulmonary nodules
Introduction
Thoracic imaging has made significant progress during the transition from film-based systems to digital radiography, including the development of more refined modalities such as dual energy subtraction (DES) imaging. However, the full potential of DES and its value in the clinical setting have yet to be fully elucidated.
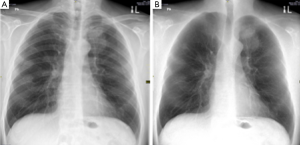
DES is an imaging technique which takes advantage of the fact that calcium selectively attenuates lower energy photons in an X-ray beam and, as a result, produces two separate images: one with the soft tissues of the thorax and one with the bones (1). There are two separate techniques that are used to obtain DES images: single exposure and double exposure.
In a single exposure system, the X-ray is exposed to two phosphor plates separated by a copper filter. The front plate receives the unfractionated beam and produces a standard chest X-ray. The lower energy photons are then selected out by the front plate and copper filter such that the back plate only receives the high energy photons. Weighed subtraction of the second image from the first is then used to produce the soft-tissue and bone selective images. In a dual exposure system, two images are taken 200 ms apart, one at 60 kV and 120 kV. The resulting images are subtracted to produce the tissue selective and bone selective images. The dual exposure system produces a higher quality image but runs the risk of motion artifacts due to the time delay between the two images that need to be adjusted in post-processing (1).
Regardless of the type of DES imaging, the modality results in two images: a soft tissue selective image and a bone selective image (Figure 1). By eliminating rib and clavicular shadows from the lung parenchyma, DES has the potential to improve the diagnosis of soft-tissue pathologies when compared to regular chest X-rays (2). There have been multiple studies that showed an increase in diagnostic accuracy of DES imaging as compared to traditional chest X-rays but none that have explored the difference in speed of diagnosis (3-6). The purpose of this study was to build on previous research by comparing the speed and accuracy of the diagnosis of non-calcified pulmonary nodules with DES imaging and traditional chest X-rays.
Materials and methods
Database
Chest radiographs of patients who underwent DES and traditional radiography at the same time between January 2011 and March 2013 were selected based on the following inclusion criteria: (I) DES radiography, chest X-ray and chest CT were done within three months of each other; (II) the presence of one or two non-calcified pulmonary nodules was confirmed by the CT; (III) the nodules were visible on both the traditional and DES radiographs; (IV) patients did not undergo any treatment for the nodules between the time of DES and CT. Based on these criteria, 34 patients were chosen (32 with one nodule, 2 with two nodules in close proximity to one another). For the control group, 17 patients were chosen who did not have any CT evidence of chest nodules or diseases but still underwent traditional and DES chest radiography.
In total, 51 patients (34 abnormal and 17 normal) were selected. The traditional chest X-ray and soft-tissue DES radiograph of each patient were then obtained, for a total of 102 images.
Software creation
Specialized image display software was created by an engineer at the University of Waterloo to allow the radiologists’ response times and accuracy to be recorded. Each image was classified by a staff radiologist as ‘normal’ or ‘abnormal’ and the locations of the nodules on the abnormal images were then inputted into the software. The abnormal images that had two nodules in close proximity were programmed such that clicking on either nodule was considered correct.
Performance testing
Five staff radiologists and five radiology residents were recruited to take part in this study. The radiology residents were all past their third year of residency training.
The readings of the images were done in two sessions that were at least 24 hours apart. In their first session, each participant read 51 images (25 DES and 26 traditional) and in their second session, they read the remaining 51 images (26 DES and 25 traditional). No participant saw both the DES and traditional X-ray of the same patient in the same session. This was done to ensure that the images were read independently.
When run, the software showed each image to the observer and asked them to identify if there was a nodule (Figure 2). They could either click a ‘No nodule’ button at the bottom of the screen if they thought the image was normal or they could click on the nodule itself, if they saw one. Once they made their decision, they clicked on the ‘Display image’ button at the bottom of the screen to proceed to the next image.
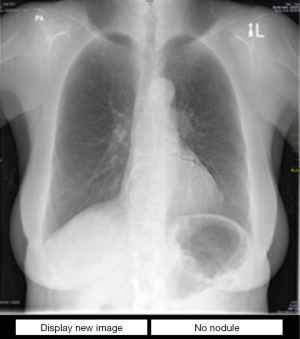
The software recorded the time it took for the radiologists to make their diagnostic decision by measuring the time from when the image was shown to them to the time they clicked ‘Display image’ to see the next radiograph. The software also recorded their diagnosis. Given that the images were being displayed on custom software, no manipulations of the level and width of the window were possible.
The readings were all done on the same monitor located at the Medical Imaging Informatics Research Centre at McMaster University. All participants were given the same set of instructions; they were informed of the objective of the study and how to use the software. No patient information was provided. At the end of each session, the data containing the participant’s diagnosis time and accuracy for each image was added to a central database. When all ten participants had completed both of their sessions, the database was analyzed by our statistician.
Analysis
For each participant, we computed the sensitivity (called positive/total positive), specificity (called negative/total negative) and accuracy (total accurate calls/total films). We also calculated the average time per read in seconds.
Each variable (sensitivity, specificity, accuracy) was then analyzed with a repeated measures ANOVA, using film (n=51) as the unit of analysis, and with three repeated measures [dual vs. single energy, resident vs. faculty, and rater (five levels)].
Results
The results are shown in Table 1.

Full table
The dual beam procedure resulted in an approximately 10% increase in sensitivity and 7% increase in overall accuracy; both had borderline significance (There was no difference in specificity. Staff readings were slightly more sensitive than residents; again the F-test was of borderline significance. The dual beam procedure reduced read times for residents by 6 sec (29%) and for staff by 3 sec (23%), both statistically significant.
Discussion
The study showed that the dual beam strategy led to a statistically and clinically significant reduction in read times, with no loss of accuracy, and a suggestion that the sensitivity to detect abnormal lesions was higher. A decrease in read times, with no decrease in accuracy, can help accommodate a larger volume of images.
One of the limitations of this study was the small sample size of images. One consequence is that the apparent increase in sensitivity did not reach statistical significance. This could be improved upon by involving other centers that use DES technology to provide a larger group of more diverse images. Secondly, due to the preliminary nature of the study and limited resources, the number of readers was limited. A multicenter study would enable participation from a larger group of radiologists and radiology residents.
DES imaging does have some disadvantages related to the technology’s high cost and difficult accessibility (7). At present, it is only located in specialized large centers. When compared with regular chest X-rays, DES technology results in twice as much radiation. However, the radiation dosage provided by a chest X-ray is 0.1 mSv and DES X-ray is 0.2 mSv, both of which are insignificant dosages considering the average Canadian is exposed to 1.77 mSv of radiation from natural sources in the course of a year (8).
Other studies have shown that DES imaging improves accuracy when compared to chest X-rays. Li et al. evaluated the accuracy and confidence of radiologists in diagnosing pulmonary nodules on 19 previously missed lung cancer nodules and showed an increase in both factors (4). Uemura et al. echoed similar results, showing an increase in accuracy in the reading of DES imaging of 52 patients with pulmonary nodules as compared to regular chest X-rays (9). While most of these studies evaluate radiologic accuracy objectively, their measure of radiologic confidence is subjectively done via scales and ratings. This study aimed to evaluate not only the accuracy of DES imaging but also the time to diagnosis, both as an independent outcome and as a surrogate outcome for confidence.
The diagnosis of non-calcified pulmonary nodules can be challenging on regular chest X-rays, particularly if the nodules are small or if they are located in more inconspicuous areas such as behind the ribs or clavicles. Shah et al. reported a series of missed malignant pulmonary nodules on chest X-ray; of the 40 total, 65% were obscured by two or three bones, 30% were obscured by one bone and 22% were obscured by the clavicle or one or more ribs (10). The benefit of DES imaging is that it eliminates overlying bone shadows by separating the soft tissues from the bones, producing two distinct images.
Given the relative novelty of DES imaging, it has yet to find its niche in currently radiologic practice. While its high cost and specialized technology makes it difficult to replace chest X-rays, there is a role for DES technology in particular populations. With the growing pool of evidence that it can be superior in the diagnosis of pulmonary nodules, it can be used to evaluate patients who are at high risk for lung cancers. This includes patients with a significant smoking history, occupational exposures, family history or symptoms concerning for a lung malignancy. It can also be used to evaluate patients with other malignancies who are at a risk of lung metastases.
Conclusions
DES is a budding new technology that has yet to find its niche in Radiology. While not widely used, there is a growing pool of evidence to suggest that it improves confidence and accuracy in the diagnosis of pulmonary nodules. Further research into the technology involves multicenter studies to accrue a larger sample size and more diversity within the images as well as a larger number of readers. Currently, there are studies looking into the efficacy of DES in diagnosing other chest pathologies such as mediastinal masses and bone disease in the ribs.
Acknowledgements
The authors of this paper would like to thank Jane Castelli for her support throughout the research process and the Medical Imaging Informatics Research Centre at McMaster University for allowing us to use their resources.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Kuhlman JE, Collins J, Brooks GN, Yandow DR, Broderick LS. Dual-energy subtraction chest radiography: what to look for beyond calcified nodules. Radiographics 2006;26:79-92. [PubMed]
- Gilkeson RC, Sachs PB. Dual energy subtraction digital radiography: technical considerations, clinical applications, and imaging pitfalls. J Thorac Imaging 2006;21:303-13. [PubMed]
- Kelcz F, Zink FE, Peppler WW, Kruger DG, Ergun DL, Mistretta CA. Conventional chest radiography vs dual-energy computed radiography in the detection and characterization of pulmonary nodules. AJR Am J Roentgenol 1994;162:271-8. [PubMed]
- Li F, Engelmann R, Doi K, MacMahon H. Improved detection of small lung cancers with dual-energy subtraction chest radiography. AJR Am J Roentgenol 2008;190:886-91. [PubMed]
- MacMahon H. Improvement in detection of pulmonary nodules: digital image processing and computer-aided diagnosis. Radiographics 2000;20:1169-77. [PubMed]
- Muhm JR, Miller WE, Fontana RS, Sanderson DR, Uhlenhopp MA. Lung cancer detected during a screening program using four-month chest radiographs. Radiology 1983;148:609-15. [PubMed]
- Ide K, Mogami H, Murakami T, Yasuhara Y, Miyagawa M, Mochizuki T. Detection of lung cancer using single-exposure dual-energy subtraction chest radiography. Radiat Med 2007;25:195-201. [PubMed]
- Canada Nuclear Safety Commission. Radiation Doses. Available online: http://www.nuclearsafety.gc.ca/eng/resources/radiation/introduction-to-radiation/radiation-doses.cfm, accessed 02/08 2014.
- Uemura M, Miyagawa M, Yasuhara Y, Murakami T, Ikura H, Sakamoto K, Tagashira H, Arakawa K, Mochizuki T. Clinical evaluation of pulmonary nodules with dual-exposure dual-energy subtraction chest radiography. Radiat Med 2005;23:391-7. [PubMed]
- Shah PK, Austin JH, White CS, Patel P, Haramati LB, Pearson GD, Shiau MC, Berkmen YM. Missed non-small cell lung cancer: radiographic findings of potentially resectable lesions evident only in retrospect. Radiology 2003;226:235-41. [PubMed]


