Endovascular stroke treatment now and then—procedural and clinical effectiveness and safety of different mechanical thrombectomy techniques over time
Introduction
More than 30 years after the first case of endovascular stroke treatment (EST) was reported, five prospective studies have established EST as the preferred treatment option for acute ischemic stroke (AIS) due to large vessel occlusion (LVO) in the anterior circulation (1-6). While intra-arterial thrombolysis and mechanical thrombus fragmentation have been the sole interventional treatment options for a long time, the advent of endovascular thrombectomy devices in the early years of the new millennium was a milestone in EST (7,8). After the introduction of various devices such as the MERCI® device (Concentric, Mountain View, USA), the Phenox clot retriever (Phenox, Bochum, Germany), or the Penumbra® system (Penumbra, Alameda, USA), stent-retrievers such as the SolitaireTM device (ev3, Irvine, USA) emerged as the most effective thrombectomy devices (7-10). While complete recanalization was also possible with the first endovascular thrombectomy devices, recanalization could be very time-consuming and was not always associated with favorable clinical outcome (11). Modern stent-retrievers, however, allow for rapid, effective, and safe revascularization of the occluded vessels, which in turn is associated with favorable clinical outcome (2-6). This is why the use of modern stent-retrievers is one of the keys to successful stroke treatment. Purpose of this study was to evaluate whether the introduction of modern stent-retrievers as first-line the treatment device resulted in faster procedural times and whether a significantly lower number of interventional techniques per case were needed.
Methods
Patients
Our study is based on a retrospective analysis of two EST cohorts that have been exclusively treated by the same interventionalist in two different hospitals. The first cohort consisted of 36 consecutive patients, who received EST in a maximum care hospital (HELIOS, Schwerin, Germany) between April 2008 and January 2010. The second cohort consisted of 50 consecutive patients who received EST in a university hospital (RWTH Aachen University, Aachen, Germany) between March 2011 and September 2014. While the first cohort was analyzed retrospectively, the analysis of the second cohort is based on a prospective stroke registry.
The following criteria were used to select patients eligible for endovascular treatment: (I) treatment was possible within 6 hours of symptom onset; (II) there was LVO in CT angiography; (III) perfusion CT showed a mismatch between cerebral blood flow and cerebral blood volume, indicating salvageable “penumbra” brain tissue of at least 20% of the infarcted area. In some cases, patients with more than 6 hours of symptom onset were considered also, if all of the following criteria were met: (I) neurological symptoms were severe; (II) the area of irreversibly damaged brain was small; (III) the area showing a mismatch as described above was large. Patients in both cohorts were included in this study regardless of occlusion site and specific interventional treatment modality. Twenty patients in the first cohort and fifty-seven patients in the second cohort fulfilled these inclusion criteria. Only patients, for whom the clinical endpoint (modified Rankin scale, mRS, after 90 days) was available, were included in this study. This is why seven patients in the second cohort, who were lost during follow up, were excluded from this analysis.
Procedures
In order to rule out that interindividual differences of experience and technical preference of various interventionalists affect our results, we analyzed all procedures that were performed by one interventionalist (MW), who already had more than 6 years’ experience with EST before the first treatment included in this analysis. Choice of the initial recanalization method was at the discretion of the interventionalist. If complete reperfusion could not be achieved with the initial method, further techniques could be applied (“escalation”). Escalation was undertaken at the discretion of the interventionalist if an improvement of the recanalization result could be expected and additional risks were judged as being tolerable. All interventions were performed under general anesthesia.
First cohort: standard EST techniques comprised intra-arterial thrombolysis, manual aspiration of the thrombus with a microcatheter (intracranial thrombus) or with an access catheter (extracranial thrombus), as well as use of a Penumbra system and a Phenox clot retriever as described in the literature (8). Other EST techniques were stenting of the occluded intracranial vessel [M1 segment of the middle cerebral artery (MCA) or basilar artery] and stenting of extracranial stenoses [internal carotid artery (ICA) or common carotid artery (CCA) carotid artery, or vertebral artery]. EST with a Solitaire stent-retriever was considered as a salvage maneuver, whenever the previous techniques failed to revascularize the occlusion site.
Second cohort: EST with stent-retrievers was the treatment option of first choice in all cases. Second, intra-arterial thrombolysis and third, stenting of the occluded vessel were considered whenever the previous treatment option failed to revascularize the occlusion site.
Clinical and radiological data
After obtaining permission from both local ethics boards, we assessed demographics (age, sex), clinical presentation (NIHSS, national institute for health stroke scale) and disability (mRS) at admission, and disability on follow-up (mRS on day 90). Radiological and procedural data comprised initial cerebral imaging with site of LVO, type of EST and devices, procedural time intervals, and result of recanalization (TICI, thrombectomy in cerebral infarction) (12,13). Hemorrhagic events were reported according to the European cooperative acute stroke study (ECASS) definition (14). Symptomatic hemorrhage was defined as radiologically proven hemorrhage accompanied by a clinical deterioration (≥4 points in the NIHSS) that could not be attributed to other causes. Procedural time intervals were analyzed for “onset to image” [onset of symptoms to first image of CT or magnetic resonance imaging (MRI) study], “image to groin” (first image of CT or MRI study to groin puncture), “groin to revascularization” (groin puncture to revascularization of target vessel), and “onset to revascularization” (onset of symptoms to revascularization of target vessel). Primary outcome measures were successful revascularization (TICI ≥2b), good functional outcome defined as mRS ≤2, and mortality on follow-up (90 days). Secondary outcome measures were defined as EST duration (time from groin puncture to recanalization) and number of applied EST techniques.
Statistical analysis
Student’s t-test, Mann-Whitney U test, Pearson χ2, and Fisher’s exact test were used depending on type of data and data distribution. P values under the alpha level of .05 were defined as significant. All statistical analyses were performed with SPSS 23 software (IBM, Armonk, New York, USA).
Results
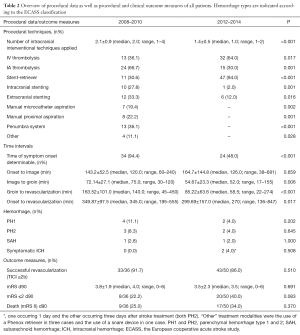
An overview of clinical, radiological, and procedural data can be found in Tables 1 and 2. Baseline characteristics such as age and sex distribution as well as stroke severity at admission and site of vessel occlusion did not differ significantly between the two cohorts (Table 1). In both cohorts, high revascularization rates (TICI ≥2b) were achieved (91.7% and 86.0%, respectively). As expected, type of treatment differed significantly between the groups (Table 2). EST with a stent-retriever was performed in 47 of 50 cases (94.0%) of the second cohort, while it was performed as a second-line treatment in 11 of 36 cases (30.6%) of the first cohort. A significantly higher number of intracranial interventional techniques per case were required in the first cohort as compared to the second cohort (Table 2). Accordingly, when only mechanical revascularization techniques were considered and cases with intra-arterial thrombolysis were excluded from this analysis, significantly fewer different intracranial EST techniques were needed in the second cohort (mean ± SD, 1.1±0.3; median, 1.0; range, 1–2) compared to the first cohort (mean ± SD, 1.7±0.7; median, 2.0; range, 1–4) (P=0.011). Significant differences were also found in in-house procedural times, with faster image-to-groin and groin-to-recanalization times in the second cohort (Table 2). The rate of patients achieving good functional outcome (mRS ≤2) was higher in the second cohort (22.2% for cohort 1 vs. 40.0% for cohort 2), although this effect did not reach statistical significance (P=0.083). Notably, all patients with a favorable clinical outcome had been successfully revascularized (TICI ≥2b). Neither in the first cohort nor across cohorts, functional outcome (P=0.699 and P=0.235), mortality (P=0.531 and P=0.380), and recanalization rates (P=0.230 and P=0.247) depended on whether stent-retrievers were used or not.

Full table
Full table
In the first cohort, there was symptomatic re-occlusion of implanted stents in two cases (9.1%). One occlusion was located in the cervical carotid artery (WALLSTENTTM, Boston Scientific, Marlborough, Massachusetts, USA) and the other was located in the MCA (SolitaireTM stent). When thrombus removal with a stent-retriever was intended, there was one case of vessel perforation with consecutive subarachnoid hemorrhage (SAH) each in the first cohort (2.8%) and in the second cohort (2%) (P=1.000). SAH was accompanied by a small cerebral hematoma (ECASS PH1) in the case from the first cohort. Hemorrhage rates are summarized in Table 2. Hemorrhage rates in the first cohort were higher, but did not differ significantly between the two cohorts (Table 2).
Discussion
EST has been subject to substantial changes in the last decade. The most essential development was the shift from sole intra-arterial thrombolysis to endovascular thrombectomy with dedicated thrombectomy devices (7,8). Our results support the hypothesis that EST with stent-retrievers as first-choice devices improves procedural times. Complete or almost complete vessel recanalization (TICI ≥2b) with acceptable complication rates was achievable in the vast majority of cases from the beginning (91.7%). However, similar to the MERCI trial before the introduction of modern stent-retrievers, this high recanalization rate was not yet associated with a satisfactory rate of good clinical outcome (22.2%) (15). When stent-retrievers became the device of first choice, recanalization was achieved almost twice as fast (85 vs. 163 minutes on average) (Table 2). EST not only became faster, but also more efficient. The number of different EST techniques needed for vessel recanalization (including IA thrombolysis) dropped from 2.1 to 1.4 (Table 2). These procedural improvements were also reflected in improved clinical outcome. When stent-retrievers became the device of first choice, the proportion of patients with favorable functional outcome (mRS ≤2) on follow-up almost doubled (40.0% vs. 22.2%). Even though this effect failed to reach statistical significance, this trend shows that optimization of EST results in more favorable clinical outcome. One might object that the clinical outcome in the first cohort was not as good as might be expected in the first place. Yet, when comparing our results to the recent prospective studies, one has to bear in mind that we included a wider range of patients for mechanical recanalization (2-6). In contrast to the recent prospective studies we included patients with occlusions in the posterior circulation and proximal occlusions of the carotid artery. Nonetheless, our results must be interpreted with great care, given the retrospective approach of our study. In fact, our data do not support the hypothesis that the use of stent-retrievers per se results in a more favorable outcome. One must also bear in mind that EST with stent-retrievers was not the treatment option of first choice and that the rate of systemic thrombolysis was significantly lower in the first cohort. Consequently, our results imply in more general terms that overall optimization of EST with an effective thrombectomy technique—which is the use of stent-retrievers in our case—results in improved procedural and clinical outcome.
Limitations
The major limitation of our study is the relatively small sample size and the retrospective approach, both of which provoke speculative interpretation of our data to some extent. As the cohorts were treated in different hospitals, infrastructural factors might also have an impact on the results. Also, one cannot certainly rule out the impact of a personal learning curve, even though the interventionalist had more than 6 years’ experience with EST before the first treatment included in this analysis.
Conclusions
Full revascularization of the target vessel was achievable before modern stent-retrievers were established as the treatment device of first choice. However, a significantly lower number of interventional techniques per case were required and recanalization was achieved almost twice as fast when stent-retrievers were established as first-line the treatment device.
Acknowledgements
None.
Footnote
Conflicts of Interest: Grants: Stryker Neurovascular, Siemens Healthcare; Personal fees: Stryker Neurovascular, Silkroad Medical, Siemens Healthcare, Bracco. And other authors have no conflicts of interest to declare.
Ethical Statement: This study was approved by our local ethics committee (registration number: EK 335/15) and written informed consent was obtained from all patients.
References
- Zeumer H, Hündgen R, Ferbert A, Ringelstein EB. Local intraarterial fibrinolytic therapy in inaccessible internal carotid occlusion. Neuroradiology 1984;26:315-7. [PubMed]
- Goyal M, Demchuk AM, Menon BK, Eesa M, Rempel JL, Thornton J, Roy D, Jovin TG, Willinsky RA, Sapkota BL, Dowlatshahi D, Frei DF, Kamal NR, Montanera WJ, Poppe AY, Ryckborst KJ, Silver FL, Shuaib A, Tampieri D, Williams D, Bang OY, Baxter BW, Burns PA, Choe H, Heo JH, Holmstedt CA, Jankowitz B, Kelly M, Linares G, Mandzia JL, Shankar J, Sohn SI, Swartz RH, Barber PA, Coutts SB, Smith EE, Morrish WF, Weill A, Subramaniam S, Mitha AP, Wong JH, Lowerison MW, Sajobi TT, Hill MD, ESCAPE Trial Investigators. Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med 2015;372:1019-30. [Crossref] [PubMed]
- Jovin TG, Chamorro A, Cobo E, de Miquel MA, Molina CA, Rovira A, San Román L, Serena J, Abilleira S, Ribó M, Millán M, Urra X, Cardona P, López-Cancio E, Tomasello A, Castaño C, Blasco J, Aja L, Dorado L, Quesada H, Rubiera M, Hernandez-Pérez M, Goyal M, Demchuk AM, von Kummer R, Gallofré M, Dávalos A,. REVASCAT Trial Investigators. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N Engl J Med 2015;372:2296-306. [Crossref] [PubMed]
- Saver JL, Goyal M, Bonafe A, Diener HC, Levy EI, Pereira VM, Albers GW, Cognard C, Cohen DJ, Hacke W, Jansen O, Jovin TG, Mattle HP, Nogueira RG, Siddiqui AH, Yavagal DR, Baxter BW, Devlin TG, Lopes DK, Reddy VK, du Mesnil de Rochemont R, Singer OC, Jahan R, SWIFT PRIME Investigators. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N Engl J Med 2015;372:2285-95. [Crossref] [PubMed]
- Berkhemer OA, Fransen PS, Beumer D, van den Berg LA, Lingsma HF, Yoo AJ, Schonewille WJ, Vos JA, Nederkoorn PJ, Wermer MJ, van Walderveen MA, Staals J, Hofmeijer J, van Oostayen JA. Lycklama à Nijeholt GJ, Boiten J, Brouwer PA, Emmer BJ, de Bruijn SF, van Dijk LC, Kappelle LJ, Lo RH, van Dijk EJ, de Vries J, de Kort PL, van Rooij WJ, van den Berg JS, van Hasselt BA, Aerden LA, Dallinga RJ, Visser MC, Bot JC, Vroomen PC, Eshghi O, Schreuder TH, Heijboer RJ, Keizer K, Tielbeek AV, den Hertog HM, Gerrits DG, van den Berg-Vos RM, Karas GB, Steyerberg EW, Flach HZ, Marquering HA, Sprengers ME, Jenniskens SF, Beenen LF, van den Berg R, Koudstaal PJ, van Zwam WH, Roos YB, van der Lugt A, van Oostenbrugge RJ, Majoie CB, Dippel DW; MR CLEAN Investigators. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med 2015;372:11-20. [Crossref] [PubMed]
- Campbell BC, Mitchell PJ, Kleinig TJ, Dewey HM, Churilov L, Yassi N, Yan B, Dowling RJ, Parsons MW, Oxley TJ, Wu TY, Brooks M, Simpson MA, Miteff F, Levi CR, Krause M, Harrington TJ, Faulder KC, Steinfort BS, Priglinger M, Ang T, Scroop R, Barber PA, McGuinness B, Wijeratne T, Phan TG, Chong W, Chandra RV, Bladin CF, Badve M, Rice H, de Villiers L, Ma H, Desmond PM, Donnan GA, Davis SM. EXTEND-IA Investigators. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med 2015;372:1009-18. [Crossref] [PubMed]
- Nogueira RG, Schwamm LH, Hirsch JA. Endovascular approaches to acute stroke, part 1: Drugs, devices, and data. AJNR Am J Neuroradiol 2009;30:649-61. [Crossref] [PubMed]
- Grunwald IQ, Wakhloo AK, Walter S, Molyneux AJ, Byrne JV, Nagel S, Kühn AL, Papadakis M, Fassbender K, Balami JS, Roffi M, Sievert H, Buchan A. Endovascular stroke treatment today. AJNR Am J Neuroradiol 2011;32:238-43. [Crossref] [PubMed]
- Nogueira RG, Lutsep HL, Gupta R, Jovin TG, Albers GW, Walker GA, Liebeskind DS, Smith WS. TREVO 2 Trialists. Trevo versus Merci retrievers for thrombectomy revascularisation of large vessel occlusions in acute ischaemic stroke (TREVO 2): a randomised trial. Lancet 2012;380:1231-40. [Crossref] [PubMed]
- Broussalis E, Trinka E, Hitzl W, Wallner A, Chroust V, Killer-Oberpfalzer M. Comparison of stent-retriever devices versus the Merci retriever for endovascular treatment of acute stroke. AJNR Am J Neuroradiol 2013;34:366-72. [Crossref] [PubMed]
- Gobin YP, Starkman S, Duckwiler GR, Grobelny T, Kidwell CS, Jahan R, Pile-Spellman J, Segal A, Vinuela F, Saver JL. MERCI 1: a phase 1 study of Mechanical Embolus Removal in Cerebral Ischemia. Stroke 2004;35:2848-54. [Crossref] [PubMed]
- Fugate JE, Klunder AM, Kallmes DF. What is meant by "TICI"? AJNR Am J Neuroradiol 2013;34:1792-7. [Crossref] [PubMed]
- Pexman JH, Barber PA, Hill MD, Sevick RJ, Demchuk AM, Hudon ME, Hu WY, Buchan AM. Use of the Alberta Stroke Program Early CT Score (ASPECTS) for assessing CT scans in patients with acute stroke. AJNR Am J Neuroradiol 2001;22:1534-42. [PubMed]
- Trouillas P, von Kummer R. Classification and pathogenesis of cerebral hemorrhages after thrombolysis in ischemic stroke. Stroke 2006;37:556-61. [Crossref] [PubMed]
- Smith WS, Sung G, Saver J, Budzik R, Duckwiler G, Liebeskind DS, Lutsep HL, Rymer MM, Higashida RT, Starkman S, Gobin YP, Multi MERCI Investigators. Frei D, Grobelny T, Hellinger F, Huddle D, Kidwell C, Koroshetz W, Marks M, Nesbit G, Silverman IE. Mechanical thrombectomy for acute ischemic stroke: final results of the Multi MERCI trial. Stroke 2008;39:1205-12. [Crossref] [PubMed]


