Classifying thoracolumbar fractures: role of quantitative imaging
Introduction
Almost 90% percent of all spinal injuries involve the thoracolumbar (TL) region (1). More than 50% of thoracic and lumbar fractures occur between T11 and L1 (2), while 25–40% of the factures affect the rest of the dorsal spine and 10–14% the rest of the lumbosacral spine (3). Fifty percent of TL fractures are unstable and can result in significant anatomic injury and deformity (4). Neurological deficit is present in 20–40% of TL fractures with most paraplegics sustaining trauma between the T11 to L2 spinal segment (4,5).
Clinical assessment of patients with TL fractures is often challenging and, as a result, diagnostic imaging usually plays an essential role in their exact diagnosis and appropriate management (6). The aim of this article is to review the role of different imaging methods in studying TL fractures, emphasizing the role of the radiologist in classifying and quantifying the severity of these fractures.
Imaging TL fractures: which technique should I use?
Radiography
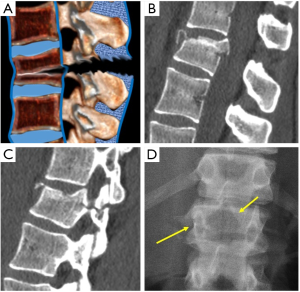
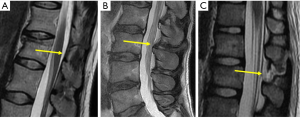
Radiographs are the adequate starting modality for patients who have sustained a low-energy trauma. AP and lateral views are usually performed. Both projections are useful in assessing vertebral height and the presence of fracture lines. The AP view allows the measurement of the interpedicular distance, which is increased in burst fractures, and the interspinous distance, which is increased in posterior ligamentous complex (PLC) injuries. These values can be reported as millimeters or as a percentage relative to adjacent normal levels. Regarding interspinous distance, variations of up to 7 mm are considered as normal (7). Calculation of the percentage of the spinous processes widening compared with adjacent normal levels is also useful, with 20% of widening being considered as sign of an unstable PLC, requiring surgical treatment (Figures 1,2) (8).
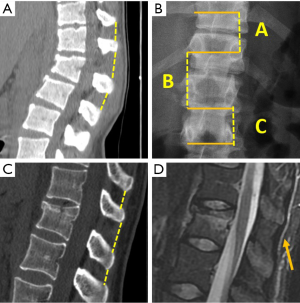
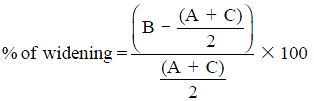 . (C) Sagittal MPR in MDCT shows 7 mm interspinous widening suspicious of PCL tear that was ruled out by MRI (arrow in D).
. (C) Sagittal MPR in MDCT shows 7 mm interspinous widening suspicious of PCL tear that was ruled out by MRI (arrow in D).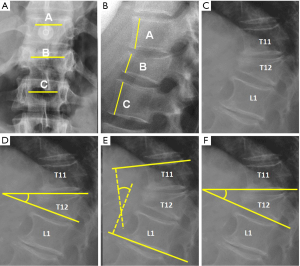
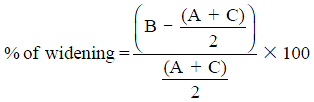 ; (B) Anterior vertebral height. The percentage of vertebral height loss can be calculated by the following formula, where A is the height of normal superior vertebra, B is the height of the fractured vertebral body and C is the height of normal inferior vertebra:
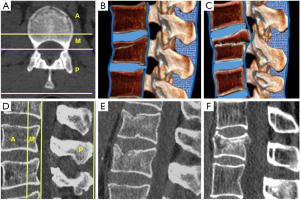
; (B) Anterior vertebral height. The percentage of vertebral height loss can be calculated by the following formula, where A is the height of normal superior vertebra, B is the height of the fractured vertebral body and C is the height of normal inferior vertebra: 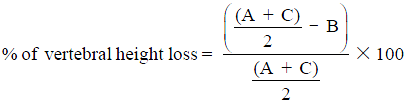 ; (C) Wedge fracture of T12; (D) local Kyphosis is the angle between both endplates of fractured vertebra; (E) regional kyphosis is the angle between the upper endplate of the vertebra overlying the fractured vertebral body and the lower endplate of the vertebra underlying the fractured vertebral body; (F) segmental kyphosis (SK) is the angle between the inferior endplate of the injured vertebra and the inferior endplate of the overlying vertebra (segment = injured vertebra + overlying disc).
; (C) Wedge fracture of T12; (D) local Kyphosis is the angle between both endplates of fractured vertebra; (E) regional kyphosis is the angle between the upper endplate of the vertebra overlying the fractured vertebral body and the lower endplate of the vertebra underlying the fractured vertebral body; (F) segmental kyphosis (SK) is the angle between the inferior endplate of the injured vertebra and the inferior endplate of the overlying vertebra (segment = injured vertebra + overlying disc).On lateral radiographs the two main parameters to be measured are vertebral height loss and kyphotic deformity (6) (Figure 2). Kyphosis is the most common deformity observed in TL spine fractures and there are several ways to quantify it. Local vertebral kyphosis angle is measured between the tangent to the upper endplate and the lower endplate of the injured vertebra. We have to be reminded that vertebral wedging is not always synonymous of vertebral fracture. In normal children and adults, the vertebral body is anteriorly wedged from T1 through L2 (peak at T7), non-wedged at L3, and posteriorly wedged at L4 to L5 (peak at L5) (9). The superior limits of normal wedging have been reported in the literature as a ratio between the anterior and posterior vertebral height. In asymptomatic adults, this limit can reach 10º (10) and 11º in children (11), although other schemes broaden this limit up to 20º–25º (12,13). Nevertheless, although fractures without vertebral deformity or wedging can occur, a vertebral height reduction >15% is considered by other authors as one of the morphometric criteria required for radiographic diagnosis of an incident vertebral fracture (14). This variability indicates a lack of consensus on the exact definition of an osteoporotic vertebral fracture by spinal radiography. These cutoff points are used to avoid the inclusion of other non-fractures entities which lead to reduced vertebral body height in the absence of fracture, such as physiological wedging, short vertebral height (SVH), Scheuermann’s disease-Schmorl’s nodes, degenerative scoliosis or Cupid’s bow deformity (15). Another morphological criterion for osteoporotic vertebral fracture is the presence of a concave depression of the endplate. Therefore, a reduction greater than 15% in the anterior vertebral height without endplate depression is categorized as non-osteoporotic SVH, which includes normal variation in height or developmental abnormalities (16).
Orthopedic surgeons may also make treatment decisions based on other measurements, such as regional kyphosis or the sagittal index. Regional kyphosis is the angle defined by the tangent to the upper endplate of the vertebra overlying the fracture and the tangent to the lower endplate of the vertebra underlying the injured vertebra. This is the recommended method by the Spine Trauma Group Study in quantifying kyphotic deformity due to its higher reliability (17).
The sagittal index (SI) is defined as segmental kyphotic deformity minus the baseline sagittal contour in the segment with the fractured vertebral body. The segmental kyphosis is the angle between the inferior endplate of the injured vertebra and the inferior endplate of the overlying vertebra. The baseline sagittal contour in each vertebral segment arbitrarily amounts to +5° for the thoracic region, 0° T12–L1 and −10° for the lumbar spine segments. The normal index is 0 (18).
Multidetector computed tomography (MDCT)
In most centers, spine CT is nowadays included as the starting modality in the imaging protocol of high energy trauma, not only if back pain is present, but in all cases as part of ruling out bone fractures and associated thoracoabdominal injuries. Long bone fractures and traumatic brain injury can act as distracting injuries, and therefore, total spine CT is mandatory in these cases. In poly-trauma patients, demonstration of a vertebral fracture in a segment of the spine is indication of scanning the whole spine with CT because up to 20% of these patients show non-contiguous vertebral fractures (19).
MDCT is also indicated in cases of low energy trauma with normal radiographs, if clinical suspicion persists. A tailored CT scan covering the painful segment is recommended due to the low sensitivity of radiography for detecting vertebral fractures, ranging from 33–77%, according with the affected level, being lower in the upper thoracic spine and higher in the lumbar spine (6,20). When fractures are demonstrated on radiographs of patients with low energy trauma, we advocate to perform a CT scan including at least two vertebrae up and below the fracture, because it is well known that radiographs underestimate the severity of fractures, including instability, or misdiagnose burst fractures as anterior compression fractures (21,22). Therefore, radiographs alone cannot be used for surgical planning and additional CT scanning is mandatory for accurate fracture classification and treatment-decision making (23).
All measurements performed by plain film radiographs can be reproduced to better advantage by MDCT. Measurements of canal dimensions are also more accurate. The sagittal-to-transverse canal diameter ratio, the canal total cross-sectional area, and the percent of canal stenosis are considered the most useful parameters in predicting neurological damage (17) (Figure 3). Comparison is performed with the pre-injury canal diameter calculated from the intact levels above and below the fractured vertebra. The ratio of sagittal to transverse diameter at the level of the injury has been shown to be significantly decreased in patients with neurologic deficit, mainly due to increase of the interpedicular transverse diameter that leads to a more ellipsoid vertebral canal shape. A ratio under 0.40 is seen in most of the patients with neurologic symptoms (24). Smaller cross-sectional areas can be tolerated at caudal levels without neurologic deficit. Previous research has concluded that the percentage of canal stenosis needed for neurologic compromise varies according localization. Significant risk is present when canal narrowing is ≥35% at T11 to T12, ≥45% at L1 and ≥55% at L2 and below (25).
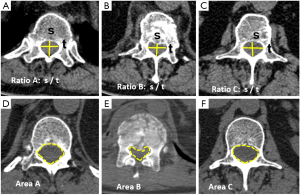
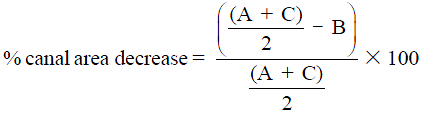 .
.In fracture-dislocation the degree of translation of vertebral body is also measured. Vertebral translation greater than 3.5 mm has been reported to be associated with PLC injury (7).
The sensitivity of CT for depicting TL fractures ranges from 95–100% (26,27). It is also more accurate than magnetic resonance imaging (MRI) for detecting fractures involving the posterior elements, and for demonstration of the size and location of loose osseous fragments (27).
Magnetic resonance imaging
Although neurological status is usually assessed clinically, MRI is recommended to determine the exact location and extent of the damage. From a radiological point of view medullary lesions are graded into three types based on T2 weighted images: (I) representing cord hemorrhage, shows initial hypointensity on MRI and prognosis is poor; (II) representing cord edema, shows initial hyperintensity and have the best prognosis; and (III) considered a contusion or small central hemorrhage surrounded by edema, shows a mixed pattern and intermediate prognosis (28). MRI can also quantify the extent of anatomical injury. Spinal cord edema confined to 1 vertebral segment or less has a much better prognosis for neurological recovery than extending over a longer segment (29).
Because PLC injury, even in the absence of neurological damage, may also indicate surgery, MRI is needed when clinical exam or CT do not clearly assure the integrity of the PLC. Rupture of the ligaments is depicted as frank interruption of a normally dark ligament replaced by high signal intensity fluid. MR imaging accuracy has been reported to be higher for detecting supraspinous ligament and ligamentum flavum injuries, and slightly lower for interspinous ligament and facet capsular injuries (30).
Types of traumatic vertebral fractures
Quantification of TL fracture severity based on imaging is paramount for accurate classification into fracture type and appropriate treatment guidance. Denis’ three columns model is helpful for defining the basic types of fractures (31). Compression fractures are characterized by an isolated failure of the anterior column. Therefore, the posterior vertebral wall and the spinal canal are intact (Figure 4).
Burst fractures are the result of compression mechanisms or as part of a hyperflexion-extension or rotation injury (32). The anterior and middle column are disrupted secondary to axial loading. Its radiographic signs are disruption of the posterior vertebral body wall, loss of the posterior vertebral height with retropulsion of the posterior vertebral body margin into the canal, and an increased interpedicular distance (Figure 4).
In flexion-distraction fractures all three columns are affected. Distraction means separation of two parts, the middle and posterior column, with the anterior column acting as a pivot. This mechanism is associated with a high incidence of intra-abdominal injuries. Their typical radiographic findings include interspinous widening, transverse fractures through the pedicles and/or other posterior elements and increased height of the posterior vertebral body and/or posterior intervertebral disc. On the AP view, interspinous widening is shown as the “empty vertebral body sign” (33) (Figure 5).
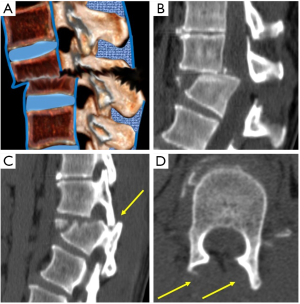
Fracture-dislocation injuries are usually the result of multidirectional forces, including compression and/or distraction in combination with some degree of shear or rotation (34). The diagnosis can be made with radiographs based on the observation of vertebral displacement of dislocated facets, although CT shows to better advantage the displacement and canal stenosis. Dislocated facets can also be demonstrated on axial images by analyzing the naked facet secondary to the loss of the normal relationship between facets (35). These fractures are extremely unstable and are associated with the highest incidence of neurologic injury (Figure 6).
Non-traumatic vertebral fractures
With population ageing only 14% of the fractures are traumatic, while most of the fractures (86%) are due to low energy trauma: 83% followed moderate or no trauma in conditions of general fragility of bone, mainly in osteoporotic patients, being classified as insufficiency fractures, and 3% are pathologic, secondary to osseous involvement of a focal lesion, mainly tumors (36).
Low energy fractures can be classified as insufficiency fractures or pathologic fractures. When this is not possible, imaging guided biopsy may be indicated. In addition, even when imaging indicates a pathologic fracture, biopsy is still needed. Several radiological signs have been described to support this differential diagnosis, with MRI playing the main role in this task due to its capacity to detect fractures before radiographic morphologic changes appear (14).
Acute osteoporotic vertebral fractures tend to show a band-like pattern of subchondral edema and, quite often, the linear pattern of the vertebral fracture can be depicted inside the edema. A retropulsed bone fragment and the presence of intra vertebral cleft are characteristic of benign compression fractures. Chronic vertebral compression fractures are characterized by morphologic changes with recovery of normal signal of the bone marrow (Figure 7) (37).
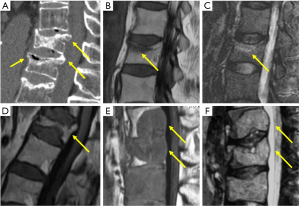
Pathologic fractures may show complete substitution of normal bone marrow or, when incomplete, tend to show and nodular or patchy pattern. Morphologic signs are a convex vertebral border, due to vertebral cortex expansion by a growing tumor, and the presence of an asymmetric paravertebral mass (Figure 7) (38).
Classification systems
To decide management, accurate classification of the fracture is critical. Understanding the historical evolution of classification systems is useful in achieving this purpose. Before X-ray discovery in 1895, spinal fractures were classified based on the presence or absence of neurological injury (39).
Since Boehler first proposed his injury categories in 1929, after the spread of radiography use, many advances have been achieved in the understanding of fracture mechanisms, imaging and classification. These systems were based on the anatomy and mechanism of the fractures (40), instability and posterior element integrity (41,42).
Holdsworth was the first author introducing the column concept in 1963. He divided the spine into an anterior and posterior column, separated by the posterior longitudinal ligament, considering this ligament as the most important for spinal stability. Therefore, compression and burst fracture, that share the same axial load failure mechanism, were considered stable because the posterior column was not damaged (43).
The novel two columns model of Holdworth was challenged by other authors that proposed a three columns model, pinpointing the posterior vertebral wall as the mainstay of spinal stability (44,45). The most popular and widespread of these works was Denis’ three columns concept (31). The posterior column was the same as that described by Holdsworth, while the middle column included the posterior wall of the vertebral body, the posterior annulus fibrosis and the posterior longitudinal ligament. Although this system is considered useful to explain the different types of spinal fractures, it is not as successful in predicting spinal stability. In fact, the classification of all fractures with involvement of two of the three columns as unstable is considered by many as an oversimplification since it is well-known that additional criteria are needed to classify two-column burst fractures as stable or unstable. Biomechanical cadaveric studies demonstrated that the integrity of the posterior column was a far better indicator of fracture stability than the integrity of the middle column (46).
Several classifications systems contributed to the understanding of TC lumbar fractures, such as the one by Mcafee (47), deepening in the concept of instability, or the AO-Magerl system, which is comprehensive but complex (32).
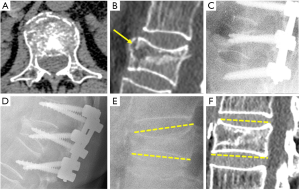
McCormack and Gaines in 1994 introduced the load sharing classification system to predict the risk of implant failure after short segment posterior fixation of TL fractures (48). The factors influencing fixation failure include the amount of vertebral body comminution on sagittal images, the apposition of bony fragments seen on axial images and the amount of kyphotic deformity correction by comparing pre- and postoperative films. Each factor scores 1 to 3 according to severity. Scores ≥7 indicate the need of longer multilevel posterior fixation or anterior vertebral body reconstruction (Figure 8). Radiologist are able to quantify comminution and apposition of bony fragments on CT images. They could also predict the capacity of kyphotic correction, even before surgery, in cases for which comparison between CT, performed in extension, and radiography, performed in the sitting, lateral decubitus or standing position, were available. Kyphotic correction is a sign of vertebral instability and may influence the type of surgical treatment (49).
The Thoracolumbar Injury Severity Score (TLISS) classification was created by the Spine Trauma Study Group in 2005 (50). Three parameters are scored in order to classify patients who require surgical or non-surgical management. These parameters are mechanism of injury, neurological status and integrity of the PLC. Nevertheless, after a study by the same group, a modification of the system was proposed in which the mechanism of injury was changed by the type of fracture in an attempt to improve intra and inter-observer reliability. It is supposed that injury morphology is easier to elucidate than having to infer the injury mechanism from static radiological studies. This new system is called the thoracolumbar injury classification and severity score (TLICS) (51).
A score <4 points is indicative of medical treatment, >4 indicative of surgical management, while if it is =4 points the decision is based on surgical modifiers. Factors that may indicate surgical management include severe local kyphosis or vertebral collapse, open fractures, obesity that precludes wearing of a brace, or, in case of poly-trauma, need of early mobilization. Factors against surgery include severe wounds or burns of the soft tissues, medical comorbidity and poor bone quality, such as in severe osteoporosis.
Fracture morphology takes into account only the most severely damaged fractures, scoring from 1, compression, to 4, distraction fracture, while burst and translational/rotational injuries score 2 and 3, respectively.
Neurological injury values range from 0 in case of normal neurological status (ASIA type E) to 3 points in case of incomplete spinal cord injury (ASIA B-D) or cauda equina syndrome, while complete spinal cord (ASIA A) or nerve root injury scores 2. Because of this component, a radiologist is unable to completely classify according to TLICS, as information on neurological injury is needed.
Posterior ligament complex injury scores 0 when intact, 2 when the lesion is doubtful or indeterminate, and 3 when the injury is evident (Figure 9). Assessment of PLC is important because its failure significantly influences the severity of the fracture (52). In a recent study MRI increased the severity of CT score of TL fractures in 31% of patients and in 22% changed the indication for conservative treatment (score <5 points) to indication for surgery (score ≥5 points) (53).
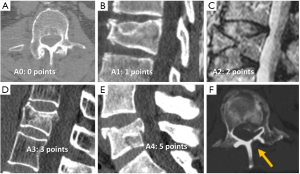
A new comprehensive modified AO classification system has recently been proposed, the AOSpine-TLICS (54). It takes the best of the Magerl and TLICS methods trying to overcome the limitations of each. The new system only scores fracture morphology and neurologic status, but relevant patient-specific modifiers are considered in therapeutic decisions. Fracture morphology is classified in 3 main types of injury: Type A is a compression injury without involvement of PLC and scores 0 (A0) to 5 (A4) (Figure 10). The more severe subtypes, A3 and A4, include vertebral body burst fractures with retropulsion of the posterior vertebral wall without disruption of the PLC and, therefore, considered stable. Fractures of the posterior arch are vertically oriented.
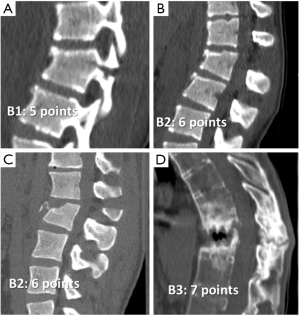
In type B injuries there is damage of so-called the tension bands; that is, the PLC or the anterior longitudinal ligament. Fracture extension through the posterior elements is horizontally oriented and disrupts the stability of the spine. They can be a monosegmental bony posterior tension band injury, B1, that scores 5, a mono or multisegmental bony and/or ligamentary failure of the posterior tension band, B2, that scores 6, or injuries of the anterior longitudinal ligament, B3, that scores 7 (Figure 11). B3 injuries occur particularly in the ankylosed spine, maintaining an intact posterior element hinge that prevent gross displacement (55). Type C is a translation/displacement injury and scores 8. Neurological injuries are scored 0 to 4 according to the severity. Assessment of PLC is now included among modifiers and score 1 when present or indeterminate on MR images. Patients with more than 5 points in this score system should undergo surgical intervention (56).
Take home messages: radiological report with quantitative information influences treatment
Findings and measurements in the radiological report should be preferably included in agreement with referring orthopedic surgeons. Radiologists need to describe the anatomy of the fracture based on the primary types of Denis’ classification (compression, burst, flexion-distraction and fracture-dislocation) (31), and finally classify it according to recent version of AOspine thoracolumbar spine Injury classification system (54). This score strongly influences therapeutic choice. Patients with more than five points in this score system should undergo surgical intervention (56). That means that all B2, B3 and C type fractures have to be managed surgically, except if there is medical contraindication. Surgical or non-surgical treatment is acceptable for A4 and B1 fractures. The rest of the fractures can be managed conservatively, except when neurological or other clinical modifiers increase score severity.
Basic radiographic measurements include the degree of vertebral wedging (local Kyphosis) and millimeters or percentage of vertebral height loss in case of compression fractures. It should be noted that loss of anterior vertebral body height >50% and local vertebral kyphotic angulation >30º–35º can be an indication for surgery (1).
In burst fractures, information about the degree of canal stenosis should be added. It can be reported as a percentage, although the influence of the degree of stenosis in neurological damage varies between vertebral levels (25). Nevertheless, a sagittal-transverse diameter ratio <0.40 is highly associated to neurological injury (24).
In more severe types of fractures, the degree of translation and widening of the interspinous distance can also be reported. Vertebral translation greater than 3.5 mm has been reported to be associated with PLC injury (7). Regarding interspinous distance, variations of up to 7 mm can be normal and 20% of widening is considered as sign of an unstable PLC, requiring surgical treatment (8).
In case for which extension CT and non-extension radiographs are available, the degree of kyphotic correction as a sign of vertebral instability can be predicted (49).
MRI is mainly indicated when doubts about damage severity and treatment decisions persist after CT evaluation of the fracture. It can correlate clinical neurological findings with the severity and extension of spinal cord damage, and the presence and severity of PLC injury (57). PCL integrity can change treatment to conservative therapy or to minimally invasive surgery (4).
Acknowledgements
None.
Footnote
Conflict of Interest: The authors have no conflicts of interest to declare.
References
- Alpantaki K, Bano A, Pasku D, Mavrogenis AF, Papagelopoulos PJ, Sapkas GS, Korres DS, Katonis P. Thoracolumbar burst fractures: a systematic review of management. Orthopedics 2010;33:422-29. [Crossref] [PubMed]
- DeWald RL. Burst fractures of the thoracic and lumbar spine. Clin Orthop Relat Res 1984.150-61. [PubMed]
- Gertzbein SD. Scoliosis Research Society. Multicenter spine fracture study. Spine (Phila Pa 1976) 1992;17:528-40. [Crossref] [PubMed]
- Rajasekaran S, Kanna RM, Shetty AP. Management of thoracolumbar spine trauma: An overview. Indian J Orthop 2015;49:72-82. [Crossref] [PubMed]
- Kelly RP, Whitesides TE Jr. Treatment of lumbodorsal fracture-dislocations. Ann Surg 1968;167:705-17. [Crossref] [PubMed]
- Anderson MW. Imaging of Thoracic and Lumbar Spine Fractures. Seminars in Spine Surgery 2010;22:8-19. [Crossref]
- Radcliff K, Su BW, Kepler CK, Rubin T, Shimer AL, Rihn JA, Harrop JA, Albert TJ, Vaccaro AR. Correlation of Posterior Ligamentous Complex Injury and Neurological Injury to Loss of Vertebral Body Height, Kyphosis, and Canal Compromise. Spine (Phila Pa 1976) 2012;37:1142-50. [Crossref] [PubMed]
- Neumann P, Wang Y, Kärrholm J, Malchau H, Nordwall A. Determination of inter-spinous process distance in the lumbar spine Evaluation of reference population to facilitate detection of severe trauma. Eur Spine J 1999;8:272-8. [Crossref] [PubMed]
- Masharawi Y, Salame K, Mirovsky Y, Peleg S, Dar G, Steinberg N, Hershkovitz I. Vertebral body shape variation in the thoracic and lumbar spine: characterization of its asymmetry and wedging. Clin Anat 2008;21:46-54. [Crossref] [PubMed]
- Matsumoto M, Okada E, Kaneko Y, Ichihara D, Watanabe K, Chiba K, Toyama Y, Fujiwara H, Momoshima S, Nishiwaki Y, Hashimoto T, Takahata T. Wedging of vertebral bodies at the thoracolumbar junction in asymptomatic healthy subjects on magnetic resonance imaging. Surg Radiol Anat 2011;33:223-28. [Crossref] [PubMed]
- Gaca AM, Barnhart HX, Bisset GS 3rd. Evaluation of wedging of lower thoracic and upper lumbar vertebral bodies in the pediatric population. AJR Am J Roentgenol 2010;194:516-20. [Crossref] [PubMed]
- Genant HK, Wu CY, van Kuijk C, Nevitt MC. Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res 1993;8:1137-48. [Crossref] [PubMed]
- Szulc P, Munoz F, Marchand F, Delmas PD. Semiquantitative evaluation of prevalent vertebral deformities in men and their relationship with osteoporosis: the MINOS Study. Osteoporos Int 2001;12:302-10. [Crossref] [PubMed]
- McKiernan FE. The broadening spectrum of osteoporotic vertebral fracture. Skeletal Radiol 2009;38:303-8. [Crossref] [PubMed]
- Griffith JF. Identifying osteoporotic vertebral fracture. Quant Imaging Med Surg 2015;5:592-602. [PubMed]
- Diacinti D, Guglielmi G. Vertebral Morphometry. Radiol Clin N Am 2010;48:561-75. [Crossref] [PubMed]
- Keynan O, Fisher CG, Vaccaro A, Fehlings MG, Oner FC, Dietz J, Kwon B, Rampersaud R, Bono C, France J, Dvorak M. Radiographic measurement parameters in thoracolumbar fractures: a systematic review and consensus statement of the spine trauma study group. Spine (Phila Pa 1976) 2006;31:E156-65. [Crossref] [PubMed]
- Farcy JP, Veidenbaum M, Glassman S. Sagittal index in management of thoracolumbar burst fractures. Spine (Phila Pa 1976) 1990;15:958-65. [Crossref] [PubMed]
- Meena S, Sharma P, Chowdhury B. Management of Thoracolumbar Fractures. Indian J Neurosurg 2015;4:56-62. [Crossref]
- Sheridan R, Peralta R, Rhea J, Ptak T, Novelline R. Reformatted visceral protocol helical computed tomographic scanning allows conventional radiographs of the thoracic and lumbar spine to be eliminated in the evaluation of blunt trauma patients. J Trauma 2003;55:665-69. [Crossref] [PubMed]
- Campbell SE, Phillips CD, Dubovsky E, Cail WS, Omary RA. The value of CT in determining potential instability of simple wedge-compression fractures of the lumbar spine. Am J Neuroradiol 1995;16:1385-92. [PubMed]
- Ballock RT, Mackersie R, Abitbol JJ, Cervilla V, Resnick D, Garfin SR. Can burst fractures be predicted from plain radiographs? J Bone Joint Surg Br 1992;74:147-50. [PubMed]
- Dai LY, Wang XY, Jiang LS, Jiang SD, Xu HZ. Plain Radiography Versus Computed Tomography Scans in the Diagnosis and Management of Thoracolumbar Burst Fractures. Spine (Phila Pa 1976) 2008;33:E548-52. [Crossref] [PubMed]
- Vaccaro AR, Nachwalter RS, Klein GR, Sewards JM, Albert TJ, Garfin SR. The significance of thoracolumbar spinal canal size in spinal cord injury patients. Spine (Phila Pa 1976) 2001;26:371-76. [Crossref] [PubMed]
- Hashimoto T, Kaneda K, Abumi K. Relationship between traumatic spinal canal stenosis and neurologic deficits in thoracolumbar burst fractures. Spine (Phila Pa 1976) 1988;13:1268-72. [Crossref] [PubMed]
- Berry GE, Adams S, Harris MB, Boles CA, McKernan MG, Collinson F, Hoth JJ, Meredith JW, Chang MC, Miller PR. Are plain radiographs of the spine necessary during evaluation after blunt trauma? Accuracy of screening torso computed tomography in thoracic/lumbar spine fracture diagnosis. J Trauma 2005;59:1410-13. [Crossref] [PubMed]
- Diaz JJ Jr, Cullinane DC, Altman DT, Bokhari F, Cheng JS, Como J, Gunter O, Holevar M, Jerome R, Kurek SJ Jr, Lorenzo M, Mejia V, Miglietta M, O'Neill PJ, Rhee P, Sing R, Streib E, Vaslef S, EAST Practice Management Guideline Committee. Practice management guidelines for the screening of thoracolumbar spine fracture. J Trauma 2007;63:709-18. [Crossref] [PubMed]
- Bondurant FJ, Cotler HB, Kulkarni MV, McArdle CB, Harris JH Jr. Acute spinal cord injury. A study using physical examination and magnetic resonance imaging. Spine (Phila Pa 1976) 1990;15:161-8. [Crossref] [PubMed]
- Schaefer DM, Flanders AE, Osterholm JL, Northrup BE. Prognostic significance of magnetic resonance imaging in the acute phase of cervical spine injury. J Neurosurg 1992;76:218-23. [Crossref] [PubMed]
- Pizones J, Izquierdo E, Alvarez P, Sánchez-Mariscal F, Zúñiga L, Chimeno P, Benza E, Castillo E. Impact of magnetic resonance imaging on decision making for thoracolumbar traumatic fracture diagnosis and treatment. Eur Spine J 2011;20:S390-6. [Crossref] [PubMed]
- Denis F. The three column spine and its significance in the classification of acute thoracolumbar spinal injuries. Spine (Phila Pa 1976) 1983;8:817-31. [Crossref] [PubMed]
- Magerl F, Aebi M, Gertzbein SD, Harms J, Nazarian S. A comprehensive classification of thoracic and lumbar injuries. Eur Spine J 1994;3:184-201. [Crossref] [PubMed]
- Bernstein MP, Mirvis SE, Shanmuganathan K. Chance-type fractures of the thoracolumbar spine: imaging analysis in 53 patients. AJR Am J Roentgenol 2006;187:859-68. [Crossref] [PubMed]
- Nagel DA, Koogle TA, Piziali RL, Perkash I. Stability of the upper lumbar spine following progressive disruptions and the application of individual internal and external fixation devices. J Bone Joint Surg Am 1981;63:62-70. [Crossref] [PubMed]
- Lingawi SS. The naked facet sign. Radiology 2001;219:366-7. [Crossref] [PubMed]
- Cooper C, Atkinson EJ, O’Fallon WM, Melton LJ. Incidence of clinically diagnosed vertebral fractures: a population based study in Rochester, Minnesota, 1985-1989. J Bone Miner Res 1992;7:221-27. [Crossref] [PubMed]
- Ruiz Santiago F, Santiago Chinchilla A, Guzmán Álvarez L, Pérez Abela AL, Castellano García Mdel M, Pajares López M. Comparative review of vertebroplasty and kyphoplasty. World J Radiol 2014;6:329-43. [Crossref] [PubMed]
- Ruiz Santiago F, Castellano García MM, Guzmán Álvarez L, Tello Moreno M. Computed tomography and magnetic resonance imaging for painful spinal column: contributions and controversies. Radiologia 2011;53:116-33. [Crossref] [PubMed]
- Burrell HL. I. Fracture of the spine: a summary of all the cases (244) which were treated at the Boston City Hospital from 1864 to 1905. Ann Surg 1905;42:481-506. [Crossref] [PubMed]
- Böhler L. Die techniek der knochenbruchbehandlung im grieden und im kriege. Vienna, Austria: Verlag von Wilheim Maudrich; 1930, 9-11.
- Watson-Jones R. The results of postural reduction of fractures of the spine. J Bone Joint Surg Am 1938;20:567-86.
- Nicoll EA. Fractures of the dorso-lumbar spine. J Bone Joint Surg Br 1949;31B:376-94. [PubMed]
- Holdsworth F. Fractures, dislocations, and fracture-dislocations of the spine. J Bone Joint Surg Am 1970;52:1534-51. [Crossref] [PubMed]
- Decoulx P, Rieunau G. Fractures of the dorsolumbar spine without neurological disorders. Rev Chir Orthop Reparatrice Appar Mot 1958;44:254-322. [PubMed]
- Roy-Camille R, Saillant G, Marie-Anne S, Mamoudy P. Treatment of vertebral fractures and dislocations of the thoracolumbar area. Orthopade 1980;9:63-8. [PubMed]
- James KS, Wenger KH, Schlegel JD, Dunn HK. Biomechanical evaluation of the stability of thoracolumbar burst fractures. Spine 1994;19:1731-40. [Crossref] [PubMed]
- McAfee PC, Yuan HA, Fredrickson BE, Lubicky JP. The value of computed tomography in thoracolumbar fractures. An analysis of one hundred consecutive cases and a new classification. J Bone Joint Surg Am 1983;65:461-73. [Crossref] [PubMed]
- McCormack T, Karaikovic E, Gaines RW. The load sharing classification of spine fractures. Spine (Phila Pa 1976) 1994;19:1741-4. [Crossref] [PubMed]
- Nakamae T, Fujimoto Y, Yamada K, Hashimoto T, Olmarker K. Efficacy of Percutaneous Vertebroplasty in the Treatment of Osteoporotic Vertebral Compression Fractures with Intravertebral Cleft. Open Orthop J 2015;9:107-13. [Crossref] [PubMed]
- Vaccaro AR, Lehman RA Jr, Hurlbert RJ, Anderson PA, Harris M, Hedlund R, Harrop J, Dvorak M, Wood K, Fehlings MG, Fisher C, Zeiller SC, Anderson DG, Bono CM, Stock GH, Brown AK, Kuklo T, Oner FC. A new classification of thoracolumbar injuries: the importance of injury morphology, the integrity of the posterior ligamentous complex, and neurologic status. Spine (Phila Pa 1976) 2005;30:2325-33. [Crossref] [PubMed]
- Harrop JS, Vaccaro AR, Hurlbert RJ, Wilsey JT, Baron EM, Shaffrey CI, Fisher CG, Dvorak MF, Oner FC, Wood KB, Anand N, Anderson DG, Lim MR, Lee JY, Bono CM, Arnold PM, Rampersaud YR, Fehlings MG. Intrarater and interrater reliability and validity in the assessment of the mechanism of injury and integrity of the posterior ligamentous complex: a novel injury severity scoring system for thoracolumbar injuries. Invited submission from the Joint Section Meeting on Disorders of the Spine and Peripheral Nerves, March 2005. J Neurosurg Spine 2006;4:118-22. [Crossref] [PubMed]
- Machino M, Yukawa Y, Ito K, Kanbara S, Morita D, Kato F. Posterior ligamentous complex injuries are related to fracture severity and neurological damage in patients with acute thoracic and lumbar burst fractures. Yonsei Med J 2013;54:1020-5. [Crossref] [PubMed]
- Winklhofer S, Thekkumthala-Sommer M, Schmidt D, Rufibach K, Werner CM, Wanner GA, Alkadhi H, Hodler J, Andreisek G. Magnetic resonance imaging frequently changes classification of acute traumatic thoracolumbar spine injuries. Skeletal Radiol 2013;42:779-86. [Crossref] [PubMed]
- Vaccaro AR, Oner C, Kepler CK, Dvorak M, Schnake K, Bellabarba C, Reinhold M, Aarabi B, Kandziora F, Chapman J, Shanmuganathan R, Fehlings M, Vialle L. AOSpine Spinal Cord Injury & Trauma Knowledge Forum. AOSpine thoracolumbar spine injury classification system: fracture description, neurological status, and key modifiers. Spine (Phila Pa 1976) 2013;38:2028-37. [Crossref] [PubMed]
- Novella Navarro M, Muñoz Gómez MM, Santiago FR, Álvarez ER. Uncommon localization of pathologic vertebral fracture in ankylosing spondylitis. Joint Bone Spine 2015;82:371. [Crossref] [PubMed]
- Vaccaro AR, Schroeder GD, Kepler CK, Cumhur Oner F, Vialle LR, Kandziora F, Koerner JD, Kurd MF, Reinhold M, Schnake KJ, Chapman J, Aarabi B, Fehlings MG, Dvorak MF. The surgical algorithm for the AOSpine thoracolumbar spine injury classification system. Eur Spine J 2016;25:1087-94. [Crossref] [PubMed]
- Rajasekaran S, Vaccaro AR, Kanna RM, Schroeder GD, Oner FC, Vialle L, Chapman J, Dvorak M, Fehlings M, Shetty AP, Schnake K, Maheshwaran A, Kandziora F. The value of CT and MRI in the classification and surgical decision-making among spine surgeons in thoracolumbar spinal injuries. Eur Spine J 2016. [Epub ahead of print]. [Crossref] [PubMed]


